-
Patient Profile: A 68-year-old male ischemic stroke survivor, 6 weeks post-event, non-ambulatory with right-sided weakness, dependent on caregiver support for daily activities.
-
Primary Clinical Challenges: Severe motor weakness, reduced voluntary movement, impaired short-term memory, fragmented sleep (3–4 hours per night), and slowed, effortful speech.
-
Chronic Comorbid Conditions: Long-standing hypertension, type 2 diabetes mellitus, and stable chronic cardiovascular disease requiring continuous pharmacological management.
-
Ongoing Standard-of-Care Medications: The patient remained on prescribed antihypertensive agents (ACE inhibitors or ARBs, calcium channel blockers, beta-blockers when indicated), antidiabetic therapy (metformin, DPP-4 inhibitors, SGLT2 inhibitors or basal insulin), and cardiovascular protection (antiplatelet agents and statins) throughout the recovery program.
-
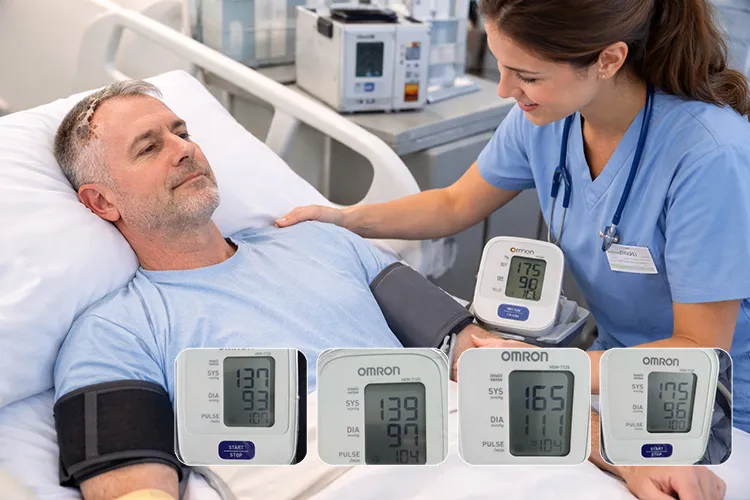
Baseline Clinical Metrics: Blood pressure 152/92 mmHg, heart rate 88 bpm, fasting blood glucose 142 mg/dL, postprandial glucose 198 mg/dL, estimated HbA1c ~7.6%, and oxygen saturation (SpO₂) 96%.
-
Home-Based Rehabilitation Approach: A gentle, bed-based rehabilitation model focusing on passive and assisted range-of-motion exercises, sensory stimulation, and controlled breathing techniques, designed specifically for non-ambulatory patients with limited cardiovascular tolerance.
-
Physiological Safety Monitoring: Daily monitoring of blood pressure, heart rate, and blood glucose was performed. Rehabilitation activities were conducted only when blood pressure remained below 150/90 mmHg and blood glucose levels were ≥ 90 mg/dL.
-
NeuroReBorn™ Clinical Ultra – Supportive Role: NeuroReBorn™ Clinical Ultra was incorporated as a non-pharmacological supportive solution to help optimize the neurological recovery environment, support cerebral circulation, and improve tolerance to light rehabilitation without replacing medical treatment.
-
Compatibility With Concomitant Medications: NeuroReBorn™ Clinical Ultra was used concurrently with antihypertensive, antidiabetic, and cardiovascular medications without requiring dose adjustment, discontinuation, or modification of existing prescriptions.
-
Safety Observations: Over an 8-week monitoring period, no hypotension, hypoglycemia, cardiac arrhythmia, excessive sedation, or clinically relevant adverse interactions were observed.
-
Clinical Outcomes After 8 Weeks: Blood pressure improved to 138/85 mmHg, heart rate stabilized at 76 bpm, fasting glucose decreased to 118 mg/dL, estimated HbA1c improved to ~7.0%, sleep duration increased to 5–6 hours per night, and early voluntary hand movement with clearer short-phrase speech was observed.
-
Clinical Interpretation: For stroke survivors with multiple chronic conditions, functional recovery is best supported through consistent, low-intensity neurological activation rather than aggressive physical training, especially in a home-based setting.
-
Conclusion: NeuroReBorn™ Clinical Ultra can be safely integrated into long-term, home-based stroke recovery programs for non-ambulatory patients with hypertension, diabetes, and cardiovascular disease, without disrupting standard medical therapy.

Safe Home-Based Stroke Recovery for Patients with Multiple Chronic Conditions

Clinical Case Study: Post-Stroke ...

Stroke Recovery: Treatment Effectiveness and ...

Post-Stroke Recovery: What Science Shows — ...

Optimizing Post-Stroke Hand Recovery with ...

Post-Stroke Functional Recovery: A 45-Day ...
Supporting Post-Stroke Recovery Through ...

A Structured, Personalized Approach to ...

A 45-Day Neuro-Rehabilitation Protocol for ...